Glycemic Index as a diet tool? Effective or Not?
Ever tried to / or thought about using the Glycemic Index as a diet tool? Whilst it is an old reference for choosing certain foods for our diets, it is still followed by many people as a means for weight loss. The below paper has been put together by our in house food scientist to assist you in making a more informed decision about its accuracy for your food consumption.
1.0 Abstract
It is generally seen that low Glycemic index (GI) foods are beneficial due to their less rise in blood glucose levels. Understanding how the GI is developed and measured leaves us with the question as to whether or not it is sufficient enough to use as a diet tool for losing weight. The hormone Insulin is seen to play a role in metabolism therefore it is importantly noted that insulin response increases at a much faster rate than do glucose response (Lee and Wolever, 1998) and there are many other substances in our body that affect insulin response. As well as understanding that, looking at a number of flaws within the development of the GI, such as the need for standardisation of table values and the various factors can alter the GI of a food, we begin to see how it may not be the most accurate tool. The concept of the Glycemic load helps to simplify the use of the GI, whereby it takes into account the amount of carbohydrates per food item, though it still does not give us an accurate understanding of the metabolic affects of foods in our body. A brief discussion of studies in relation to using the GI values as a guideline, proves the ineffectiveness of its concept with regards to a tool for losing weight.
2.0 Introduction
The glycemic index (GI) was initially conceived by (Jenkins et al., 1981) as a tool for dietary management of type 1 diabetes. Over many years it has become clear of the glycemic effects of various foods. It has been suggested that foods with a high glycemic index promote a high glycemic response, thus altering the appetite, causing an increase in hunger and consequently an accumulation of body fat. (Brand-Miller et al., 2002). In-turn a low GI diet is hypothesised to increase satiety and reduce voluntary food intake. (Nielsen et al., 2005). Thus low GI foods are generally considered beneficial due to the minimal increase in blood glucose compared to high GI foods. It is important to remember that throughout this discussion the GI was originally conceived as an inherent property of the food, not as a metabolic response of an individual to a food, thus any food is believed to have the same glycemic response (regardless of other foods in is ingested with) from person to person. So is the glycemic index a useful tool for weight loss. Beginning with an outline of the glycemic index and its calculation this paper will then discuss the glucose and insulin response and the factors that influence the GI of a food it will then outline, with support from various studies the importance of using a low GI in weight loss needing to be further studied.
3.0 Glycemic Index
Depending on what type of Carbohydrate it is, depends on how it behaves in our body. The development of the Glycemic index can help describe the different carbohydrates by ranking them according to their effect on the blood sugar levels, i.e. distinguish between good carbohydrates and bad carbohydrates. A scale of 0-100 is used depending on how much they raise blood sugar levels after eating. High GI foods are rapidly digested and result in a distinct fluctuation of blood sugar levels. Low GI foods are slowly digested and absorbed therefore produce a gradual rise in blood sugar levels. (Glycemic Index, 2007)
3.1 Measuring the GI
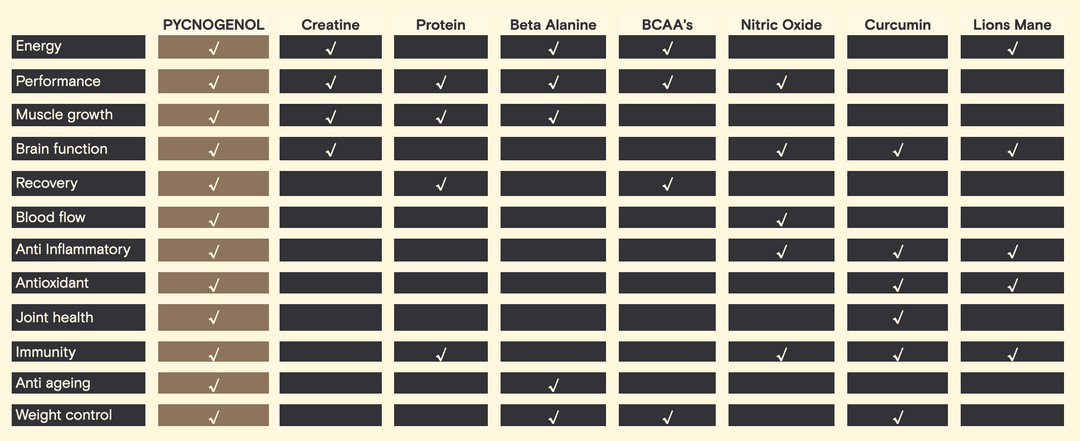
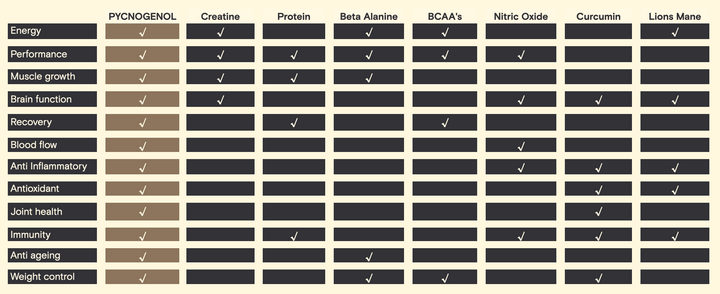
The measures taken in order to assign a GI to a food are quite strict. 10-50 gram portions of carbohydrate are fed to 10 healthy people after an overnight fast. Blood samples are then taken at 15-30 minute intervals over two hours. The blood samples are then graphed in order to create, what is known as a blood sugar response curve (for two hours). The area under the curve (AUC) is calculated to reflect the total rise in blood glucose levels after eating the test food. The GI rating (%) is calculated by
dividing the AUC for the test food by the AUC for the reference food (same amount of glucose) and multiplying by 100 (see Figure 1).
Figure 1: Area under the curve (blood glucose response curve)

The current validated reference food is glucose. This has a GI rating of 100. Though, white bread can also be used as a reference food, as many peoples diet consist of it. This changes the scale and puts glucose up to 140. Therefore there are disadvantages with this system as the reference foods are not well defined, making the GI scale culture dependent. (Brouns et al., 2005)
3.2 The need for standardised method of measurement of GI
There is a set of guidelines in order to test the GI of a food, which are:
- Tested in the morning
- Standardisation of physical activity and previous meal
- At least 10 fasting test subjects (healthy)
- 50g of available carbohydrates
- Reference product: Glucose or white bread
- Capillary blood
- Two hour incremental area
Over many years, differences in GI tests have been used, such as:
- Different blood samples: Venous and Capillary
- Different subjects: Healthy and Diabetics
- Different reference products: Glucose and Bread
Taking Bread as an example, this shows how standardising the measurement of the GI is important. There are a variety of bread products on the market which use:
- Different wheat
- Different products and;
- Different baking procedures
3.3 Glucose and Insulin response
Following a meal, blood glucose concentration rises. This stimulates insulin secretion which induces rapid entry of glucose into cells and stops glucose output by the liver, consequently reducing the concentration of glucose in the blood. Insulin then returns to its previous level, thus making it negative feedback response.
Other than just food, there are many other substances that can cause the release of insulin, these are
- Increased plasma concentration of free fatty acids
- Increase concentration of certain amino acids and
- Hormones secreted by the gastrointestinal tract in response to eating
This means that insulin secretion will rise earlier and to a greater extent than if plasma glucose was the only controller during ingestion of a meal. Exercise and stress, on the other hand, activate the sympathetic neurons and increase plasma epinephrine concentration, both of which inhibit insulin secretion.
Insulin unquestionably plays the primary role in controlling the metabolic adjustments required for feasting or fasting. Other hormonal and neural factors, glucose counter-regulatory controls, oppose the action of insulin in one way or another. The most important factor is glucagon, a peptide hormone produced by the Alpha cells of the pancreatic islets. The stimulus for glucagon secretion is a decreased plasma glucose concentration, and, conversely, an increase in plasma glucose concentration inhibits the secretion of glucagons, thereby helping to return plasma glucose level to normal.(Murray et al., 1993)
4.0 Factors affecting the GI
Eating Low GI foods may seem easy, though being aware of the array of factors that change the index rating can be quite complicated. Studies have found that present international table values are often not good predictors of measured GI for various reasons, such as differences between countries in methodology and cooking methods, indicating a need for local information. Earlier it had been assumed, based on chemical properties, that poly-saccharides or “complex carbohydrates” i.e. starch, were “slow carbo-hydrates” whereas sugars (mono- and disaccharides) were considered “rapid”. Though it is now seen that many food factors other than the molecular size of the carbohydrate component are important determinants of the glycemic response to composite foods, (Bjorck et al., 2000) even different blood glucose responses have been seen in the same weight of a carbohydrate though in different foods. (Chew et al., 1988) This would render the usefulness and practicality of the GI in diet planning.
4.1 Table Values
In tables compiled by (Foster-Powell and Miller, 1995). The Variability of glucose was 85-111. (keeping in mind, glucose is the most accurately measured carbohydrate). The data in the GI tables have been compiled over many years from various laboratories around the globe. This means the GI values have been derived from different products i.e brands, origin, different test subjects and different measurements as previously discussed. The glycemic index range follows these guidelines
- Low GI = 55 or less
- Medium GI = 56-69
- High GI = 70 or more
Table values can provide false leading information to the public. An example of this looks at carrots which due to their high GI value people tend to avoid, whereas salted peanuts are found to be excellent foods according to GI. A carrot has a GI value of 101, though to get 50g of carbohydrates from a carrot you need to eat 575g, i.e., 9 normal-sized carrots. Peanuts have a GI of 21, in order to get 50 carbohydrates from a peanut you need to eat 500g. This amount gives 2925 kcal, of which 245g are fat. This is more than the daily energy intake of most people. (Jarvi et al., 1998) These examples describe how unrealistic it can be to evaluate food as healthy or not only by its GI value. It has therefore been suggested that the GI concept should be applied only to foods providing at least 15g, and preferably 20 g, of glycemic carbohydrates per portion, i.e., products, such as bread, cereal, pasta, rice and potatoes. (Arvidsson-Lenner et al., 2004) Furthermore, comparison of GI values should generally be done within the same food groups. This prevents misunderstanding such as blacklisting carrots for example. In the literature this has also been tackled by using the concept of glycemic load (GL).
4.2 Ripeness
As a fruit ripens, starch is changed to sugar. The starch generally has a higher GI than does the sugar so as ripeness progresses, the GI decreases
Does this mean we are going to tell the public that they can eat fruit at some points in their maturation and not in others?
4.3 Physical Form of Food
Changing the particle size of some foods changes their GI. The GI of a 1-inch cube of potato can increase by 25% just by mashing the cube. Consumption of whole apples, apple purée, and apple juice results in significantly different glucose and insulin excursions. So, are we going to tell the public that they can eat one physical form of a food but not another?
4.4 Variability within food classes
The type of food, the way its processed or even the way its prepared can have an effect of the GI rating.
4.4.1 Type
Different types of foods can have a different GI. A good example of this is rice, where the proportion of amylose and amylopectin in the grains influence the GI. In order to understand this we need to be aware of the structure of amylose and amylopectin. Amylose is a linear molecule with D-glucose units linked in an (1–4) manner. Amylopectin has both (1–4) and (-1–6) linkages, (Annison and Topping, 1994) thus making it a branched structure. Amylopectin is more easily hydrolysed in the gut because of this and takes on a higher GI. (Amelsvoort and Weststrate, 1992) The international table produced by Foster-Powell demonstrated how 50g servings of different rices generate a GI range from 68 to 103. This makes selecting which variety of rice to eat very complicated, as amylose and amylopectin proportions are not stated on packaging.
4.4.2 Processing Food
Processed foods are very common in Western diets. Large scale processing conditions are very different to conventional at home methods. Grinding, rolling, pressing, or even thoroughly chewing a kernel or other starch food can disrupt the granules.
The glycemic response can be changed, depending on the way a carbohydrate is processed. It was found in a study by (Brand et al., 1985) that the more processed a food was, the higher the glycemic response. For example, rice conventionally cooked had a glycemic index of around 50, though when it is processed into an instant rice product it jumps up to 85, and further to around 93 for rice bubbles. This is quite a significant difference for both processed products. When trying to develop a diet around low GI food products for weight loss, processed foods would not generate much of a selection, as many of the products would produce a high GI rating.
4.4.3 Preparation
The type and extent of which cooking is carried out can have an affect on the GI. Heating and cooling cycles can cause starch retrogradation. For example an uncooked potato is resistant to hydrolysis , though when it is cooked the starch granules gelatinize and thus can easily be digested. If the potatoes are then cooled, the gelatinization reverses and around 12% of the potato starch becomes resistant to hydrolysis and cannot be absorbed. The amount of heat used, water and even the time of cooking also can effect the GI quite a lot. (Brand et al., 1985)
4.5 Effects of macronutrients
When we eat, our meals do not consist of just a 50g serving of carbohydrate, our meals consist of 2 or more macronutrients. Studies by (Coulston et al., 1984)
demonstrates that the higher the proportion of carbohydrate in a specific food, as opposed to protein and fat, the higher the GI; and a mixed meal of carbohydrate, protein, and fat will have a different and variable glucose response depending on the proportions of each nutrient.
4.5.1 Fats and Proteins
Fat and protein are seen to delay gastric emptying, which in turn affects insulin secretion. Their affect on the GI is not very significant unless large amounts.
- 30g Protein
- 50g Fat
This amount of fat and protein taken with 50g carbohydrates are seen to lower the GI (Wolever and Bolognesi, 1996)
4.5.2 Fibre
Wolever found that there was an inverse relation between fibre and GI when including a broad range of carbohydrate rich foods. (Wolever, 1990) It Is therefore believed that whole meal cereal products can produce a high GI the same as white bread. It has also been known for quite some time that fibre is likely to generate a range of metabolic effects. (Karlstrom et al., 1988)
4.5.3 Sugar
The GI of sucrose (using white bread as a standard) is moderately low at 65, with glucose being around 97, fructose 23 and lactose 46. (Foster-Powell and Miller, 1995) Because of fructose low GI it used much more these days in food products. The problem with the GI is that is looks at the blood glucose response, where as the metabolism of fructose takes a completely different pathway, thus having a different effect on the body. (Elliott et al., 2002) From this, fructose intake is seen to decrease satiety which can potentially lead to an increase in overall food intake, promoting weight gain. (Bray et al., 2004) This idea strongly suggest that following low GI foods can have adverse affects.
4.5.4 Acidity
Acetic and propionic acid is seen to decrease the rate of gastric emptying, where as lactic acid has shown signs of creating a barrier against enzymes that degrade starch (Darwiche et al., 2001). This in essence lowers the GI of foods, adding to another complication when diet planning. Do we need to make people aware of the amount of vinegar they can put on their food?
As we can see, there is a number of factors from processing to cooking to even food that affect the GI. This can lead consumers into false information on the tables value of GI, thus affecting diet planning.
5.0 Glycemic Load
The GI concept is only valid for food with substantial amounts of carbohydrates. This is why the glycemic load concept was developed. It takes into account the proportion of carbohydrate per food sample. Foods that are registered on the glycemic index are taken at face value by people, thus for this reason, they will avoid for eg parsnips like a plague. Though it is a fact that parsnips are mainly indigestible fibre and you would have to eat quite a few in order for it to trigger a major blood glucose rise. This is because the GI rates the effects per 10-50 grams of carbohydrate rather than the effects of per gram of the parsnip. The concept of glycemic load was introduced (Salmeron et al., 1997), aiming at giving a comparable basis of comparison that include both the quality and quantity of the carbohydrates in a food or meal. GL allows comparisons of the likely glycemic effect of realistic portions of different foods, calculated as the amount of carbohydrate in one serving times the GI of the food. For example, spaghetti has a lower GI than boiled potatoes, but the normal portion of spaghetti is commonly larger than normal portions of potatoes. (Arvidsson-Lenner et al., 2004). Therefore, GL may or may not differ between these two carbohydrate sources, depending on the applicable GI values and portion sizes.
Adopting the GL concept into diet planning would be a wise idea, although it still produces complications with regards to the factors discussed that affect the glucose response of a food. The use of GL instead of GI could also lead to a decrease consumption of carbohydrates as it would be a way to decrease the overall GL of the diet. If we look at a high GI and a low GI food with the same GL, they would not produce the same metabolic effect on the body.
6.0 Studies
As discussed earlier, insulin response is somewhat linked to glucose response. Obesity has been associated with an increase in insulin levels, therefore foods with a low GI are encouraged (Wylie-Rosett et al., 2004). Low GI foods are also believed to reduce hunger or promote satiety, thus reducing energy intake (Flint et al., 2004). It is still debated whether or not the GI is a usefull tool in weight loss. A study that keeps this debate alive, was an observation study conducted by (Ma et al., 2005). Its subject was 572 healthy adults and evaluated their body mass index over one year. The diet consisted of a low GI which often consists of foods rich in fibre. Fibre is related to increase satiety therefore the link between low GI foods and increases satiety could not be determined. This is an important issue as it is believed the power of a low GI diet is to increase satiety and ultimately reduce eating habits.
6.1 Mixed Meals
The use of the GI and its application with regards to mixed meals is debatable. It has even been concluded that GI is affected when incorporated into meals and even water is seen to increase the GI. (Hollenbeck and Coulston, 1991) In a study by (Flint et al., 2004) the main aim was to investigate the relevance of GI table values to the measured GI in breakfast meals. The study proved that the GI of mixed meals using table values did not predict the measured GI of the meal, therefore carbohydrates do not play the most important role for GI in mixed breakfast meals.
These studies demonstrate the difficulties of applying international table values to predict the GI of a specific mixed meal in our daily life. This is important to note when trying to develop a diet based on table values of low GI foods to lose weight.
7.0 Conclusion
The GI is certainly a hot topic with regards to weight loss, theoretically it suggests that it increases satiety thus reducing hunger. Though using the glycemic index with little knowledge of the factors that affect a foods rating, understanding the glycemic load and understanding the correlation a carbohydrates food may have on the insulin response does not make it an easy concept for planning a diet in order to lose weight. If the GI was a nutrient rather than a measure of the effect on blood sugar it would make use of it much easier. However, it is not a nutrient and cannot be measured like one. It takes a lot of effort to measure the GI of a single food item let alone a meal and we can see that the result is very easily affected. The importance of GI in weight maintenance or weight loss has to be further studied. It should always be kept in mind that the glycemic index is only one measure of many, which together indicate a healthy diet. It is bringing us one step closer to an answer. Until we can get a whole picture on the effect of different foods on our bodies, following a low GI diet in order to lose weight is not accurately known.
References:
AMELSVOORT, J. V. & WESTSTRATE, J. (1992) Amylose-amylopectin ratio in a meal affects postprandial variables in male volunteers. American Journal of Clinical Nutrition, 55, 712-718.
ANNISON, G. & TOPPING, D. (1994) Nutritional role of resistant starch: chemical structure vs physiological function. Annual Review of Nutrition, 14, 297-320.
ARVIDSSON-LENNER, R., ASP, N. G., AXEL-SEN, M., BRYNGELSSON, S., HAAPA, E. & JÄR-VI, A. (2004) Glycaemic index. Relevance for health, dietary recommen-dations and food labelling. Scandinavian Journal of Nutrition, 48, 84-94.
BJORCK, I., LILJEBERG, H. & OSTMAN, E. (2000) Low glycaemic-index foods. British Journal of Nutrition, 83, S149-155.
BRAND-MILLER, J. C., HOLT, S. H., PAWLAK, D. B. & MCMILLAN, J. (2002) Glycemic Index and Obesity. American Journal of Clinical Nutrition 76, 281S-285S.
BRAND, J. C., NICHOLSON, P. L., THORBURN, A. W. & TRUSWELL, A. S. (1985) Food Processing and the Glycemic Index. The Amerciann Journal of clinical nutrition 42, 1192-1196.
BRAY, G. A., NIELSEN, S. J. & POPKIN, B. M. (2004) Consumption of high-fructose corn syrup in beverages may play a role in the epidemic of obesity. American Journal of Clinical Nutrition, 79, 537-543.
BROUNS, F., BJORCK, I., FRAYN, K., GIBBS, A., LANG, V. & SLAMA, G. (2005) Glycaemic index methodology. Nutrition research reviews 18, 145-171.
CHEW, I., BRAND, J. C., THORBURN, A. W. & TRUSWELL, A. (1988) Application of glycemic index to mixed meals. American Journal of Clinical Nutrition, 47, 53-56.
COULSTON, A., HOLLENBECK, C. & REAVEN, G. (1984) Utility of studies measuring glucose and insulin responses to various carbohydrate-containing foods. American Journal of Clinical Nutrition, 39, 163-167.
DARWICHE, G., ÖSTMAN, E. M., LILJEBERG, H. G., KALLINEN, N., BJÖRGELL, O., BJÖRCK, I. M. & ALMÉR, L.-O. (2001) Measurements of the gastric emptying rate by use of ultrasonography: studies in humans using bread with added sodium propionate. American Journal of Clinical Nutrition, 74, 254-258.
ELLIOTT, S. S., KEIM, N. L., STERN, J. S., TEFF, K. & HAVEL, P. J. (2002) Fructose, weight gain, and the insulin resistance syndrome. American Journal of Clinical Nutrition, 76, 911-922.
FLINT, A., MØLLER, B. K., RABEN, A., PEDERSEN, D., TETENS, I., HOLST, J. J. & ASTRUP, A. (2004) The use of glycaemic index tables to predict glycaemic index of composite breakfast meals. British Journal of Nutrition, 91, 979-989.
FOSTER-POWELL, K. & MILLER, J. (1995) International tables of glycemic index American Journal of Clinical Nutrition, 62, 871S-893S.
HOLLENBECK, C. B. & COULSTON, A. M. (1991) The clinical utility of the glycemic index and its application to mixed meals. Candadian journal of physiology and pharmacology, 69, 100-107.
JARVI, A. E., KARLSTROM, B. & VESSBY, B. (1998) Does the glycemic index concept work in practice? Scandinavian Journal of Nutrition, 42, 83-86.
JENKINS, D., WOLEVER, T., BARKER, R. T. H., FIELDEN, H., BALDWIN, J., BOWLING, A., NEWMAN, H., JENKINS, A. & GOFF, D. (1981) Glycemic index of foods: a physiological basis for carbohydrate exchange. American Journal of Clinical Nutrition, 34, 362-366.
KARLSTROM, B., VESSBY, B., ASP, N. G. & YTTERFORS, A. (1988) Effects of four meals with different kinds of dietary fib-re on glucose metabolism in healthy subjects and non-insulin-dependent dia-betic patients. European journal of clinical nutrition 42, 519-526.
LEE, B. & WOLEVER, T. (1998) Effect of glucose, sucrose and fructose on plasma glucose and insulin responses in normal humans: comparison with white bread. European journal of clinical nutrition, 52, 924-928.
MA, Y., OLENDZKI, B., CHIRIBOGA, D., HEBERT, J. R., LI, Y., LI, W., CAMPBELL, M., GENDREAU, K. & OCKENE, I. S. (2005) Association between Dietary Carbohydrates and Body Weight American Journal of Epidemiology, 161, 359-367.
MURRAY, R. K. G., D. K, MAYES, P. A. & W., R. V. (1993) Harpers Biochemistry, 23rd Edition, Conneticut Appleton and Lange.
NIELSEN, B. M., BJØRNSBO, K. S., TETENS, I. & HEITMANN, B. L. (2005) Dietary glycaemic index and glycaemic load in Danish children in relation
to body fatness. British Journal of Nutrition, 94, 992–997.
SALMERON, J., MANSON, J. E., STAMPFER, M. J., COLDITZ, G. A., WING, A. L. & WIL-LETT, W. C. (1997) Dietary fiber, gly-cemic load, and risk of non-insulin-dependent diabetes mellitus in women. Jama, 277, 472-477.
WOLEVER, T. M. (1990) Relationship between dietary fibre content and composition in foods and the glycemic index American Journal of Clinical Nutrition, 51, 72-75.
WOLEVER, T. M. & BOLOGNESI, C. (1996) Prediction of glucose and insulin respon-ses of normal subjects after consuming mixed meals varying in energy, protein, fat, carbohydrate and glycemic index. Journal of nutrition, 126, 2807-2812.
WYLIE-ROSETT, J., SEGAL-ISAACSON, C. & SEGAL-ISAACSON, A. (2004) Carbohydrates and Increases in Obesity: Does the Type of Carbohydrate Make a Difference? Obesity research 12, 124S-129S.
GLYCEMIC INDEX, 2007, Home of the Glycemic Index, The university of Sydney, accessed 9th September 2007 from <http://www.glycemicindex.com/>

















Leave a comment